
The Fairway to Market Access - from clinical stage to pricing & reimbursement
Kompetenz für den Marktzugang von Arzneimitteln und Medical Devices
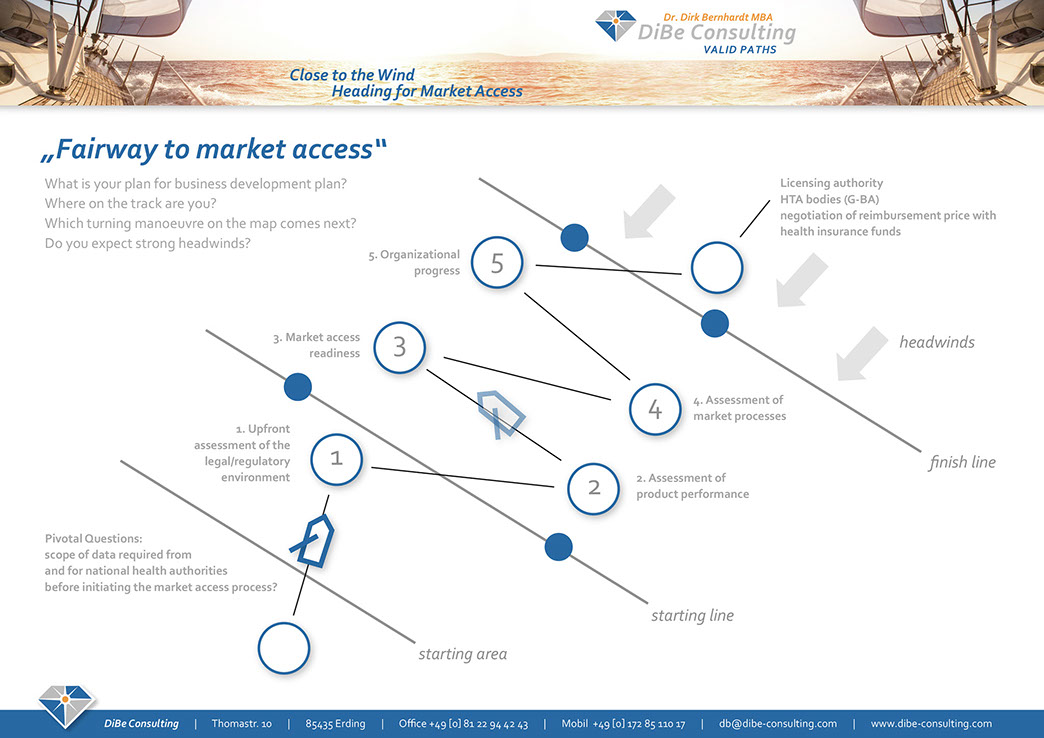
Buoy 0 – starting line
Scope of clinical data required for national health authorities before initiating the market access processes
• Study endpoints, i.e. mortality, morbidity and health-related QoL
• Study design
• Patient-related benefit of subpopulations addressed
• Nature and extent of patient-related improvement against SoC
• Disease incidence and product volume forecast
To the right of starting line
Buoy 1
Upfront assessment of the legal/regulatory environment
• Early benefit assessment according to §§35a/-137h SGB V
• New examination and treatment methods (NUB) for class IIb/III medical devices
• Readiness for AMNOG process according to §130b SGB V
Buoy 2
Assessment of Product Performance
• Nature and outcomes of treatment alternatives for the targeted indication
• Improvement over a G-BA stipulated appropriate medical intervention (zVT)
• Real world evidence reported and size of the targeted disease condition
Buoy 3
Market Access Readiness
• Evaluation of market sectors and conditions for pricing and reimbursement
• Setting of the competitive landscape i.e. provider preferences, payer constraints
• Engagement of key stakeholders, i.e. medical counselling, patient engagement
Buoy 4
Assessment of market processes
• Market size, volume sales, reimbursement, number of customers
• Outline of commercial operations, i.e. marketing & sales objectives
• COGS and profit margin
Buoy 5
Organizational progress
• Strategic imperatives and resource allocation
• Project and actions plan, i.e. launch management
• Controlling and securing the operational success
To the right of the finishing line
Licensing authority HTA bodies
(BfArM, PEI) (G-BA, IQWiG)
↓
negotiation of reimbursement price with GKV-SV
↓
Hospital procurement department Health insurance funds
(BfArM – Federal Institute for Drugs and Medical Devices │ G-BA – Federal Joint Committee │ IQWiG – Institute for Quality and Efficiency in Health Care │ PEI – Paul Ehrlich Institute │
HTA – Health Technology assessment)





International Licensing Groups
European Pharma Licensing
www.plgeurope.com
Pharmaceutical Licensing Group Limited UK (PLG)
www.plg-uk.com
Pharmaceutical Licensing Group Benelux (PLGB)
www.plgbenelux.com
Nordic Pharma Licensing Group (NPLG)
www.nplg.org
Pharma Licensing Group Spain (PLGS)
www.plgs-spain.com
Pharma Licensing Club France (FPLG)
www.plcf.org
Swiss Healthcare Licensing Group (Swiss HLG)
www.swisshlg.com
Pharmaceutical Licensing Group Italy (PLG-Italy)
www.italyhlg.it
Japanese Pharma Licensing Association (JPLA)
www.pharma-fi.com
PLG CEE Pharmaceutical Licensing Group Central and Eastern Europe
www.plg-cee.com
Canadian Healthcare Licensing Association (CHLA)
www.chlassoc.com
DiBe Consulting Member of
Pharma-Lizenz-Club Deutschland (PLCD)
www.plcd.de
Deutsche Fachgesellschaft für Market Access
www.dfgma.de
BIO Deutschland -
Branchenverband der Biotechnologie-Industrie
www.biodeutschland.org
Forum MedTech Pharma
www.medtech-pharma.de
Health Care Bayern e.V.
https://healthcare-bayern.de
https://www.novartis.de/
Market Access Medical Devices Pharma Marktzugang von Arzneimitteln weltweit
Market Access Medical Devices Pharma Marktzugang von Arzneimitteln weltweit